ISO 13485 Certification
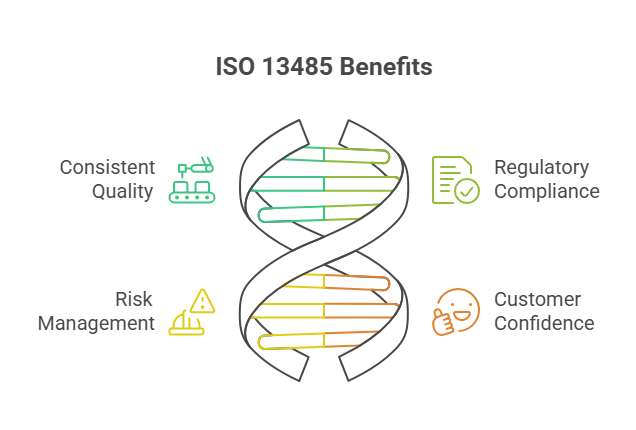
ISO 13485 certification is based on the ISO 13485 standard, which outlines the requirements for a quality management system (QMS) specific to medical device manufacturing. Unlike general quality standards, ISO 13485 focuses on risk management, process control, and regulatory alignment throughout the product lifecycle. This certification ensures that organizations consistently produce medical devices that meet both customer expectations and regulatory requirements, improv... moreISO 13485 Certification
ISO 13485 certification is based on the ISO 13485 standard, which outlines the requirements for a quality management system (QMS) specific to medical device manufacturing. Unlike general quality standards, ISO 13485 focuses on risk management, process control, and regulatory alignment throughout the product lifecycle. This certification ensures that organizations consistently produce medical devices that meet both customer expectations and regulatory requirements, improving patient safety and trust.
https://iasiso-asia.com/TH/iso-13485-certification-in-thailand/